Online Pet Vet Pharmacy: Best Deals Up to 40% Off + FREE Shipping on Orders $100+




Niglum Solution for Horses, cattle, pigs 100 ml
$64.67 Original price was: $64.67.$61.04Current price is: $61.04.
| ACTIVE INGREDIENT | Flunixin, Flunixin meglumine |
|---|---|
| HEALTH CONDITION | Anti-inflammatory |
| ANIMALS | Cattle, Horses, Pigs |
| BRAND | Calier |
| SIZE | 100ml (3.38 oz) |
| FORM | Solution, Suspension ❙ Emulsion ❙ Liquid |
Niglumine, Flunixin 50 mg/ml Solution for injection, 100 ml
Description
Transparent colorless solution with a slightly yellowish tinge.
Storage
1 ml of the drug contains the active substance:
flunixin meglumine 83.0 mg
(equivalent to 50.0 mg of flunixin).
Excipients: propylene glycol (E1520), diethanolamine, phenol, edetate disodium, sodium formaldehyde sulfoxylate, hydrochloric acid, water for injections.
Pharmacological properties
PBX classification code: C) M01AO90 Non-steroidal anti-inflammatory and anti-rheumatic drugs.
Flunixin meglumine acts as a non-selective inhibitor of cyclooxygenase (COX), which is an enzyme that converts arachidonic acid into unstable cyclic endoperoxides, which in turn are converted to prostaglandins, prostacyclins and thromboxanes. Some of them, such as prostaglandins, are involved in the pathophysiological mechanisms of inflammation (pain, fever). Decreased production of inflammatory mediators causes analgesic, antipyretic and anti-inflammatory effects of flunixin.
After intramuscular injection, flunixin is rapidly absorbed and penetrates all organs and tissues, the maximum concentration in the blood occurs within a few minutes. Despite the short half-life (average 2-8 hours), the drug accumulates at the site of inflammation, providing a therapeutic effect for 24 hours. After intravenous administration, the clinical effect of the drug is manifested in 15-30 minutes. Flunixin is 99% bound to blood proteins and excreted mainly in fecal masses, the rest – by the kidneys.
Application
Used as an anti-inflammatory, analgesic and antipyretic for complex treatment together with the etiotropic therapy of cattle, horses and pigs in acute inflammation and pain.
As an adjuvant antipyretic for the treatment of cattle with respiratory diseases (including pulmonary emphysema), mastitis, diseases
limbs, eye diseases, pathological conditions that are accompanied by acute inflammation.
Horses drug is used in diseases of the musculoskeletal system, colic to control inflammation and pain.
In the complex treatment of pigs Niglumin together with antibiotic therapy is used in MMA syndrome (metritis-mastitis-agalactia) to reduce pain.
Dosage
Cattle are administered intravenously at a dose of 2 ml of the drug per 45 kg of body weight once a day in combination with antibiotic or rehydration therapy. If necessary, the dose is repeated at 24-hour intervals for 3 days.
Horses are administered intravenously at a dose of 1 ml of the drug per 45 kg of body weight once a day in combination with antibiotic or rehydration therapy. If necessary, the dose is repeated at 24-hour intervals for 5 days.
Pigs are administered intramuscularly at a dose of 2 ml of the drug per 45 kg of body weight once a day and, if necessary, repeatedly with an interval of 12 hours, a maximum of 2 injections according to clinical manifestations and antibiotic therapy. The maximum volume of one injection should not exceed 5 ml.
*Kilograms to Pounds conversion table
| Kilograms (kg) | Pounds (lb) | Pounds+Ounces (lb+oz) |
| 0.1 kg | 0.220 lb | 0 lb 3.527 oz |
| 1 kg | 2.205 lb | 2 lb 3.274 oz |
| 5 kg | 11.023 lb | 11 lb 0.370 oz |
| 10 kg | 22.046 lb | 22 lb 0.740 oz |
Contraindication
- Do not use the drug for the treatment of animals with chronic forms of musculoskeletal disorders.
- Do not use in animals with heart, liver or kidney disease.
- Do not use in animals at risk of gastrointestinal bleeding, haemostasis disorders.
- Do not use in animals with hypersensitivity to the components of the drug.
- Do not use in cows for 48 hours until expected calving.
- Do not use in pregnant sows and foals.
- Do not use in mares whose milk is used in the food industry.
- Do not use in animals with hypotension, dehydrated or hypovolaemic animals.
- Do not use if blood dyscrasia is suspected.
- Do not use for the treatment of animals with symptoms of colic caused by intestinal torsion.
Side effect
Possible side effects include irritation of the mucous membrane of the digestive tract, ulceration, nausea, papillary necrosis of the kidneys, ataxia and pulmonary hyperventilation.
Kidney damage is possible when using the drug in animals in a state of dehydration and hypovolemia.
In pigs, local redness may appear at the injection site, which disappears on its own after the breeding period or within 28 days.
Flunixin can cause kidney damage in hypovolemic and hypotensive animals during surgery.
As with other non-steroidal anti-inflammatory drugs (NSAIDs), there is a risk of rare specific renal or hepatic adverse reactions.
Anaphylactic reactions have sometimes been fatal.
In case of any adverse reactions, the use of the drug should be stopped and consult a veterinarian immediately.
Special precautions for use
The use of the drug in animals under 6 weeks of age or older animals may pose an additional health risk. If such use cannot be avoided, the care of such animals must be very careful.
NSAIDs that inhibit prostaglandin synthesis should not be used until complete recovery in animals under general anesthesia.
In rare cases, shock reactions may occur after intravenous administration due to the content of propylene glycol. Therefore, Niglumin should be administered slowly and it should have the body temperature of the animal. At the first sign of incompatibility, the administration should be stopped and, if necessary, shock treatment should be started.
It is necessary to limit the activity of the animal during treatment with Niglumin and provide sufficient drinking water.
Intra-arterial injections should be avoided in horses and cattle. Accidental intra-arterial administration may result in ataxia, incoordination, pulmonary hyperventilation, nervousness and weakness. All these symptoms are temporary and disappear after a few minutes without the use of an antidote.
Ponies are more sensitive to the side effects of NPP, so they should be used with caution.
Cattle need to determine the cause of the inflammatory process and prescribe appropriate comprehensive treatment.
It should be borne in mind that flunixin reduces the clinical signs of inflammation in cattle and resistance to treatment (antibiotic therapy) may be hidden.
In horses, it is necessary to determine the cause of colic and prescribe appropriate comprehensive therapy.
The safety of the drug for stallions and breeding boars has not been proven. The decision to use the drug is made by a veterinarian taking into account the benefit / risk ratio.
Use during pregnancy and lactation
Do not use in cows for 48 hours until expected calving.
Do not use in pregnant sows and foals.
Interaction with other means
Do not mix with other veterinary drugs.
Do not use concomitantly with glucocorticoids, other nonsteroidal anti-inflammatory drugs and anticoagulants.
Flunixin may reduce the effect of some antihypertensive drugs, diuretics (ACE inhibitors), angiotensin receptor antagonists and B-blockers.
Avoid concomitant use with nephrotoxic drugs, especially with aminoglycosides.
Withdrawal period
Cattle: meat – 4 days, milk – 24 hours.
Horses: meat – 28 days. Do not use in mares whose milk is used in the food industry.
Pigs: meat – 28 days. .
Until this time, the products are disposed of or fed to unproductive animals, according to the conclusion of a veterinarian.
Warnings for persons and service personnel
At self-injection with the drug it is necessary to seek medical help.
Wash hands after use.
If the drug gets on the skin and mucous membranes, it is necessary to wash it off with plenty of water, if irritation occurs – seek medical attention.
Keep out of reach of children.
Do not allow people with hypersensitivity to flunixin to work with the drug.
Release form
Transparent glass vial type.II (EF), volume 100 ml, closed with a gray rubber stopper made of bromobutol (technology PH 4001/45), aluminum capsule with a ring that breaks off when opened, enclosed in a cardboard pack.
Transparent glass vial type II (EF), volume 250 ml, closed with a pink rubber stopper of bromobutol and silicate, a capsule of gold color, enclosed in a cardboard pack.
Storage
Dry dark, inaccessible to children at a temperature of 2 to 25 ° C. Do not freeze!
Shelf life – 5 years.
After the first withdrawal from the vial, the drug should be used within 28 days.
For use in veterinary medicine!
Questions and answers of the customers
You may also like…
Dexa INJ for dogs, cats, cattle, horses, sheep, goats, pigs – 100ml
Dexamethasone 2 mg/ml 100 ml INJECTED for cattle, sheep, goats, cows, pigs, horses, dogs, cats, etc.
For use:
- In inflammatory conditions of the skeletal-muscular system such as arthritis, tendinitis,
tendovaginitis, synovitis;
- Allergic skin conditions;
- Allergic lung conditions such as bronchial asthma.
Dexamethasone is a synthetic glucocorticoid with a strong anti-inflammatory effect.
The pharmacological action and therapeutic indications of the active substance are good
familiar.
As maintenance therapy, dexamethasone can be used to treat various
rheumatic, allergic, dermatological and disease-related diseases known to occur
influenced by anti-inflammatory corticosteroids. The dexamethasone solution may be
administered intravenously when immediate action is required.
Traum Solution Inj or Oral For cats, dogs, cows, horses 25ml (5ml*5pcs)
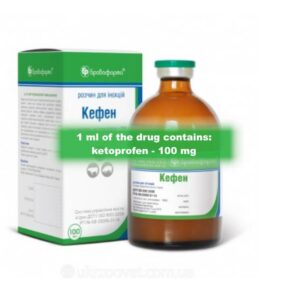
Kefan 10% for farm animals *inj (100 ml)
Kefan 10% (ketoprofen) (Ketofen analog)
Ketoprofen is a substance from the group of carboxylic acids, derivatives of propionic acid. The mechanism of action is to inhibit the synthesis of prostaglandins and thromboxane and is based on disruption of arachidonic acid metabolism. In therapeutic doses, ketoprofen inhibits mainly COX-II and partially COX-I, providing anti-inflammatory, analgesic, antipyretic and antiendotoxic effects.
After a single intramuscular injection, the maximum concentration of ketoprofen in blood plasma is observed after 30 minutes. Ketoprofen is excreted mainly by the kidneys.
Indication
Treatment of cattle, pigs, horses in acute or chronic inflammatory processes of the musculoskeletal system (arthritis, osteoarthritis, bursitis, dislocation, edema, tendovaginitis, trauma, infectious lesions of the hooves), respiratory organs, metritis-mastitis-agalactia, various forms of mastitis, pain of various etiologies (surgery, trauma, colic). Used as an antipyretic in diseases accompanied by hyperthermia and depression.
Solution-ACD 2F, 100 ml (3.38oz)
Related products
Meat flavor Tablets for dogs, 100 mg / 20 tablets
Metak 0.5% Solution for Dogs 50 ml
Chewable Tablets for Dogs #16 tablets
Meat flavor Tablets for dogs, 20 mg / 20 tablets
Firmaks 225 mg for Dogs 55-100 lbs 20 tablets
Chewable Tablets for Dogs #16 tablets
Rima 20 mg flavored tablets for Dogs 20 tablets (EXP:06/2022)
Rimadyl P 20 mg (carprofen) for dogs
A new generation anti-inflammatory drug, which is intended for the treatment of inflammatory and painful phenomena caused by acute and chronic diseases of the musculoskeletal system, as well as an analgesic and anti-inflammatory agent to reduce postoperative pain and edema in dogs.