Bamoc (Sinulox RTU analog) Amoxicillin, clavulanic acid Solution for injection, 100ml
Amoxicillin, clavulanic acid injected suspension for Cattle, Pigs, Dogs, and Cats
Description
Suspension of whitish or yellow cream color.
Storage
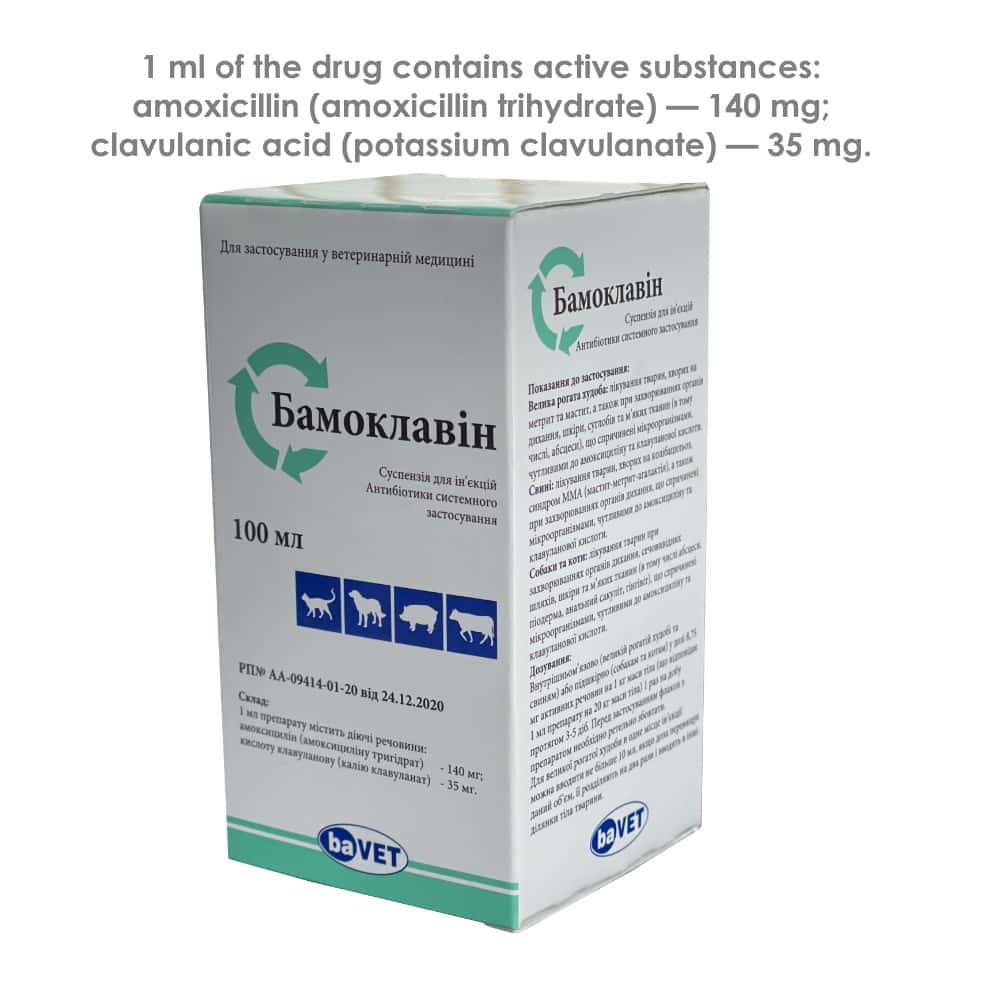
1 ml of the drug contains active substances:
amoxicillin (amoxicillin trihydrate) — 140 mg;
clavulanic acid (potassium clavulanate) — 35 mg.
Excipients: butylhydroxyanisole, butylhydroxytoluene, propylene glycol.
Pharmacological properties
ATS vet classification code QJ01 — antibacterial veterinary drugs for systemic use. QJ01CR02 — Amoxicillin and enzyme inhibitor.
Amoxicillin is an antibiotic with a broad bactericidal effect from the group of aminopenicillins. It prevents the synthesis of the cell wall by inhibiting the enzymes transpeptidase and carboxypeptidase, causing a violation of the osmotic balance, which leads to the death of bacteria at the stage of their growth. The drug is active against gram-positive (Streptococcus spp., Bacillus anthracis, Actinomyces spp., Clostridium spp., Corynebacterium spp., Erysipelothrix rhusiopathiae, Listeria monocytogenes) and gram-negative (E. coli, Pasteurella spp., Haemophilus spp., Actinobacillus spp., Bordetella bronchiseptica, Fusobacterium spp., Moraxella spp., Proteus mirabilis, Salmonella spp.) microorganisms. Most species of bacteria of the genera Klebsiella and Enterobacter, as well as the genus Pseudomonas, are resistant to amoxicillin.
Clavulanic acid is one of the natural metabolites of Streptomyces clavuligerus, it has a beta-lactam ring similar to penicillin in its structure. It is a beta-lactamase inhibitor, suppressing its activity. Clavulanic acid penetrates the bacterial cell wall, binding to extracellular and intracellular beta-lactamases.
Amoxicillin in combination with clavulanic acid significantly expands the spectrum of antimicrobial action of the drug.
The table shows the MIC 50 and MIC 90 values (expressed as amoxicillin/clavulanic acid concentration) against bacterial pathogens in dogs determined in vitro.
Minimum inhibitory concentrations (MICs) of the amoxicillin/clavulanic acid combination for bacterial pathogens in dogs.
| Pathogenic | MIC50 | MIC90 | Minimum | Maximum |
| Staphylococcus intermedius | <0.5 /0.25 | <0.5/0.25 | <0.5/0.25 | 2/1 |
| B-haemolytic Streptococcus | <0.5/0.25 | <0.5/0.25 | <0.5/0.25 | <0.5/0.25 |
| Staphylococcus aureus | 2/1 | 2/1 | <0.5 /0.25 | 2/1 |
| Escherichia coli | 4/2 | 8/4 | 1/0.5 | 64/32 |
| Pasteurella multocida | <0.5/0.25 | <0.5/0.25 | <0.5/0.25 | 1/0.5 |
After intramuscular administration to cattle and subcutaneously to dogs, amoxicillin and clavulanic acid are well absorbed and distributed in body tissues. After intramuscular administration of the drug to cattle in the recommended doses once a day for five consecutive days, the following kinetic parameters were observed: for amoxicillin – C max – 1.69 μg/ml, T max – 2.67 h, AUC – 30.59 μg/ml/h and t1/2 – 15.22 h; for clavulanic acid – C max – 0.94 μg/ml, T max – 1.3 h, AUC – 3.123 μg/ml/h and t1/2 – 1.71 h.
After subcutaneous administration of the maximum recommended dose of the drug to dogs, the following kinetic parameters for amoxicillin were observed: C max – 8.66 μg/ml, T max – 1.78 h and AUC – 50.98 μg/ml/h.
Amoxicillin and clavulanic acid are excreted from the body of animals mainly with urine.
Application
Cattle: treatment of animals suffering from metritis and mastitis, as well as diseases of the respiratory system, skin, joints and soft tissues (including abscesses) caused by microorganisms sensitive to amoxicillin and clavulanic acid.
Pigs: treatment of animals suffering from colibacillosis, MMA syndrome (mastitis-metritis-agalactia), as well as respiratory diseases caused by microorganisms sensitive to amoxicillin and clavulanic acid.
Dogs and cats: treatment of animals with diseases of the respiratory system, urinary tract, skin and soft tissues (including abscesses, pyoderma, anal saculitis, gingivitis) caused by microorganisms sensitive to amoxicillin and clavulanic acid.
Dosage
Intramuscularly (cattle and pigs) or subcutaneously (dogs and cats) at a dose of 8.75 mg of active substances per 1 kg of body weight (corresponding to 1 ml of the drug per 20 kg of body weight) 1 time a day for 3-5 days Before use, the bottle with the drug must be thoroughly shaken.
For cattle, no more than 10 ml can be injected into one injection site, if the dose exceeds this volume, it is divided into two parts and injected into other parts of the animal’s body.
| Kilograms (kg) | Pounds (lb) | Pounds+Ounces (lb+oz) |
| 0.1 kg | 0.220 lb | 0 lb 3.527 oz |
| 1 kg | 2.205 lb | 2 lb 3.274 oz |
| 5 kg | 11.023 lb | 11 lb 0.370 oz |
| 10 kg | 22.046 lb | 22 lb 0.740 oz |
Contraindication
Hypersensitivity to amoxicillin and clavulanic acid.
Do not use the drug for rabbits, ants, hamsters and other small herbivores.
Do not use in animals with impaired kidney function.
Do not use simultaneously with antibiotics of the group of cephalosporins, tetracyclines, macrolides and sulfonamides.
Do not administer intravenously.
Reservation
Side effect
In some cases, a painful reaction or a local reaction in the form of swelling may be observed after the administration of the drug. Sometimes short-term digestive disorders (nausea, diarrhea) that pass on their own, allergic reactions, anaphylaxis are possible. If one of the symptoms occurs, the drug should be discontinued and symptomatic treatment should be carried out. In the event of allergic reactions, it is advisable to administer corticosteroids and adrenaline to animals.
Special precautions for use
Clavulanic acid has a high moisture sensitivity. Therefore, in order to prevent moisture from entering the vial with the drug, it is very important to use a dry syringe and needle when drawing the solution from the vial.
The use of the drug should be based on determining the sensitivity of pathogens to the drug, as well as taking into account the rules for the use of antimicrobial drugs.
Use during pregnancy and lactation
Studies on laboratory animals have not shown a teratogenic effect. The safety of the drug has not been evaluated in pregnant and lactating cows or bitches. The use of the drug is possible only in accordance with the benefit / risk assessment by a veterinarian.
Interaction with other means or other forms of interaction
When using other penicillins, a cross-reaction is possible. Penicillins enhance the effect of aminoglycosides.
Withdrawal period (grace period)
Slaughter of animals for meat is allowed 42 days (cattle) and 31 days (pigs) after the last use of the drug. Human consumption of milk is allowed 60 hours (5 milkings) after the last use of the drug. The meat and milk obtained by the specified time are disposed of or fed to non-productive animals, depending on the opinion of the veterinary medicine doctor.
Special precautions for persons and service personnel who administer the drug
Penicillins and cephalosporins can cause hypersensitivity (allergy) after contact with the drugs. Hypersensitivity to penicillins can lead to cross-reactions with cephalosporins and vice versa. Allergic reactions to these substances can be serious. It is necessary to use the drug with caution. If there was contact with the drug and you have symptoms of allergy (redness of the skin), consult a doctor. In case of accidental injection to a person, it is necessary to immediately consult a doctor, showing the leaflet-tab or the label of the drug. Itching of the face, eyes or lips, difficulty breathing are more serious symptoms and require immediate medical attention. After working with the drug, it is necessary to wash hands.
Release form
50, 100 and 250 ml glass bottles closed with rubber stoppers under an aluminum break-in.
Secondary packaging – cardboard boxes.
Storage
Dry, dark place inaccessible to children at temperatures from 4 °C to 25 °C.
Expiration date
24 months from the date of production.
After the first selection from the bottle, the drug must be used within 28 days, provided it is stored in a dark place at a temperature from 4 °C to 25 °C.
For use in veterinary medicine.