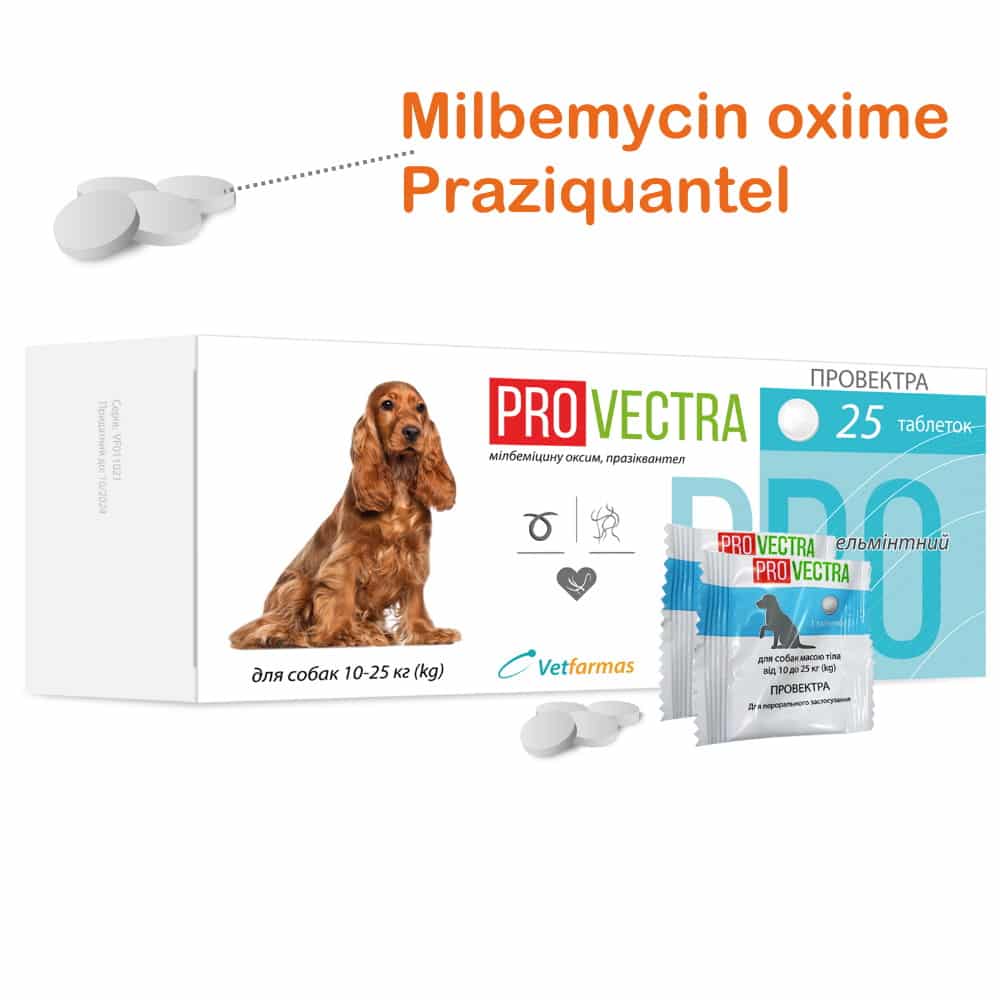
PROVECT for dogs 10-25 kg / 22-55 lb (milbemycin oxime, praziquantel), 3 tablets
Description
Round tablets are biconvex yellowish-white with brown spots.
Storage
The drug is designed for dogs with different body weights; each tablet contains the active substances at the rate of 0.5 mg of milbemycin oxime and 5 mg of praziquantel per kg of body weight of the animal.
Excipients: corn starch, lactose monohydrate, microcrystalline cellulose, povidone, magnesium stearate, colloidal silicon dioxide, meat flavoring, yeast powder.
Pharmacological properties
ATCvet QP54, endectocides (QP54AB51, milbemicini)
PROVECTRA for dogs – a combined drug with a broad spectrum of anthelmintic action. Contains active ingredients: milbemycin oxime and praziquantel.
Milbemycin oxime is a macrocyclic lactone obtained by fermentation of Streptomyces hygroscopicus, type Aureolacrimosus, active against larvae and adult nematodes that parasitize in the digestive tract of dogs, as well as against larvae of nematodes Dirofilaria immitis (microdirophylaria).
The mechanism of action of milbemycin oxime is to increase the permeability of the membranes of nematode nerve and / or muscle cells to chlorine ions, which causes their hyperpolarization. This leads to an irreversible decrease in muscle activity, paralysis and death of parasites.
The maximum concentration of milbemycin oxime in the blood plasma of dogs is reached within 2-4 hours, the bioavailability is about 80%. It is excreted from the body of animals, mostly unchanged.
Praziquantel is a derivative of acylated pyrazinisoquinoline. Praziquantel is active against cestodes and trematodes. Increasing the permeability of parasite cell membranes to calcium ions leads to depolarization of membranes, muscle contraction and damage to the tegument, which leads to the death of helminths and promotes their excretion from animals.
The maximum concentration of praziquantel in the blood plasma of dogs is reached in 1-4 hours, the bioavailability is about 80%, excreted mainly as inactive metabolites in the urine within 48 hours.
PROVECTRA for dogs according to the degree of action on the body belongs to low-toxic substances (safety class 4), in the recommended doses does not show sensitizing, embryotoxic and teratogenic effects.
Application
Prevention and treatment of dogs in invasions caused by cestodes – Dipylidium caninum, Taenia spp., Echinococcus spp., Mesocestoides spp .; nematodes – Ancylostoma caninum, Toxocara canis, Toxascaris leonina, Trichuris vulpis, Thelazia callipaeda; Angiostrongylus vasorum (reduces the intensity of infection), Crenosoma vulpis (reduces the intensity of infection), Dirofilaria immitis (disease prevention); including combined nematode-cestode invasions.
The drug can also be used to prevent heartworm disease (Dirofilaria immitis).
Dosage
The drug is administered to dogs individually once orally with food or forced to the root of the tongue in the minimum therapeutic dose: 0.5 mg of milbemycin oxime and 5 mg of praziquantel per kg of body weight.
Depending on the weight of the dog, the actual dosage is:
| Animal body weight | Dosage | ||
| milbemycin oxime | proziquantel | ||
| For very small dogs | up to 4 kg | 2 mg | 20 mg |
| For small dogs | >4 – 10 kg | 5 mg | 50 mg |
| For medium dogs | >10 – 25 kg | 12.5 mg | 125 mg |
| For large dogs | >25 – 45 kg | 22.5 mg | 225 mg |
| For very large dogs weighing more than 45 kg, the drug can be dosed with a suitable combination of tablets | |||
*Kilograms to a Pounds conversion table
| Kilograms (kg) | Pounds (lb) | Pounds+Ounces (lb+oz) |
| 0.1 kg | 0.220 lb | 0 lb 3.527 oz |
| 1 kg | 2.205 lb | 2 lb 3.274 oz |
| 5 kg | 11.023 lb | 11 lb 0.370 oz |
| 10 kg | 22.046 lb | 22 lb 0.740 oz |
Adherence to a diet or special feeding regime is not required. Separation of the tablet is not allowed.
For deworming of dogs in the invasion caused by Angiostrongylus vasorum, the drug is used for therapeutic purposes four times with an interval of 7 days; for prophylactic purposes – 1 time in 4 weeks in a therapeutic dose.
For deworming of dogs in the invasion caused by Thelazia callipaeda, the drug is used twice with an interval of 7 days.
In order to prevent heartworm disease in unfavorable regions, the drug is used in the spring-summer-autumn period monthly, starting one month before the flight of carriers of the pathogen – mosquitoes and gnats, and ending not earlier than one month after the end of their flight. Prior to deworming, consult a veterinarian to rule out the presence of microdirofilariae in the animal’s blood.
Peculiarities of the drug’s action during its first use and withdrawal have not been established.
When carrying out deworming it is necessary to adhere to the recommended terms, but not less than once a quarter. In case of skipping the next treatment, the drug is resumed in the same dose and according to the same scheme.
Contraindication
Increased individual sensitivity of animals to the components of the drug!
Severe renal and hepatic dysfunction!
Do not use in puppies under 2 weeks of age and / or weighing less than 0.5 kg!
Do not use in depleted and infectious animals!
Reservation
Deworming of animals during pregnancy and lactation is carried out as needed and under the supervision of a veterinarian.
When using the drug strictly adhere to the recommended dosage.
Collie, Bobtail and Sheltie dogs are sensitive to all anthelmintic drugs, so the drug should be used as needed and under the supervision of a veterinarian.
Treatment of dogs with high levels of circulating Dirofilaria immitis can lead to side effects (pale mucous membranes, vomiting, muscle tremors, difficulty breathing or hypersalivation). These reactions are associated with the release of proteins from dead microfilariae and do not indicate direct toxicity of the drug. Treatment of canine microfilaremia (presence of larvae in the blood) with the drug is not recommended.
In areas at risk of heartworm disease or in cases where the dog is imported or exported from the risk area, it is necessary to consult a veterinarian before using the drug to rule out the disease.
All animals kept together must be treated.
Animals weakened, sick or with impaired renal and hepatic function should be used only under the supervision of a veterinarian with a benefit / risk assessment.
Release form
PET / PE blisters of 1 tablet, packed in cardboard boxes of 2 and 25 pieces.
Storage
The drug is stored in the original packaging of the manufacturer, separately from food and feed, in a dry place, protected from direct sunlight, out of reach of children and animals at a temperature of 15 to 25 ° C.
Expiration date
3 years.
For use in veterinary medicine!