Online Pet Vet Pharmacy: Best Deals Up to 40% Off + FREE Shipping on Orders $100+


Zpril Solution for Cattle, Horse and more 100ml
$37.19 Original price was: $37.19.$33.66Current price is: $33.66.
Solution sulfamethazine + trimethoprim Zpril (Sulmet)
Sulfamethazine is a synthetic chemotherapeutic agent from the sulfamide group. Trimethoprim is a synthetic chemotherapeutic agent derived from diaminopyrimidine.
Separately, each component has a bacteriostatic mechanism, but sulfamethazine and trimethoprim, taken in a 4: 1 ratio, enhance the effect of the drug and give it a bactericidal nature of action.
Such synergy is manifested due to the fact that its components affect two sequential processes that are necessary for the synthesis of tetrahydrofolic acid or bacterial folic acid, which plays a major role in the synthesis of bacterial purines. The bacterial wall is impermeable to folates, so the synthesis of purines depends on the intrinsic synthesizing ability of microorganisms.
| ACTIVE INGREDIENT | Sulfamethazine, Trimethoprim |
|---|---|
| HEALTH CONDITION | Antibiotics |
| BRAND | INVESA, Livisto |
| ANIMALS | Camels, Cattle, Goat, Horses, Pigs, Sheep |
| FORM | Solution |
| SIZE | 100ml (3.38 oz) |
Solution sulfamethazine + trimethoprim Zpril (Sulmet)
The solution is yellowish.
Composition
1 ml of the preparation contains active ingredients:
sulfamethazine – 200 mg
trimethoprim – 40 mg.
Excipients: citric acid, sodium pyrosulfite, hydrochloric acid, sodium hydroxide, sodium methyl p-hydroxybenzonate, sodium propyl p-hydroxybenzonate, water for injection.
Pharmacological properties
Sulfamethazine is a synthetic chemotherapeutic agent from the sulfamide group. Trimethoprim is a synthetic chemotherapeutic agent derived from diaminopyrimidine.
Separately, each component has a bacteriostatic mechanism, but sulfamethazine and trimethoprim, taken in a 4: 1 ratio, enhance the effect of the drug and give it a bactericidal nature of action.
Such synergy is manifested due to the fact that its components affect two sequential processes that are necessary for the synthesis of tetrahydrofolic acid or bacterial folic acid, which plays a major role in the synthesis of bacterial purines. The bacterial wall is impermeable to folates, so the synthesis of purines depends on the intrinsic synthesizing ability of microorganisms.
These two components work as follows:
sulfamethazine, in its chemical characteristics, is similar to p-aminobenzoic acid, and together with it participates in the formation of the enzyme dihydropteroyl synthetase, counteracting the formation of dihydrofolic acid;
Trimethoprim blocks the next link in the biosynthetic chain, inhibiting the action of dihydrofolate reductase, an enzyme that accelerates the conversion of dihydrofolic acid to tetrahydrofolic acid.
The combination of these chemotherapeutic components gives the drug a bactericidal effect. At the same time, the appearance of microbial strains resistant to the drug is insignificant.
The combination of sulfamethazine-trimethoprim has a broad spectrum of action, including gram-positive and gram-negative bacteria. Microorganisms that are very sensitive to the action of the drug include the following bacteria: Escherichia coli, Clostridium spp., Shigella spp., Salmonella spp., Proteus mirabilis, Bacillus anthracis, Pasteurella spp., Haemophilus influenzae, Haemophilus gallinarum and Vibrio spp. Streptococcus pneumoniae, Streptococcus pyogenes, Streptococcus faecalis, Streptococcus viridans, Streptococcus agalactiae, Staphylococcus aureus, Proteus spp. and Brucella spp.
Application
Cattle: treatment of animals suffering from colibacillosis and salmonella gastroenteritis, vibriosis, pneumonia, septicemia, pasteurellosis, coccidiosis and colibacillosis mastitis, actinomycosis, purulent abscess, as well as diseases of the genitourinary system caused by sulphimetasis and sulphimetasis-sensitive microorganisms.
Horses: treatment of animals with diarrhea, pneumonia, pyosepticemia caused by microorganisms sensitive to sulfametazim and trimethoprim.
Pigs: treatment of animals suffering from colibacillosis, salmonellosis, vibrious dysentery, pneumonia, pasteurellosis, malignant edema, abscesses caused by microorganisms sensitive to sulfametazim and trimethoprim.
Sheep: treatment of animals suffering from colibacillosis, salmonellosis, vibrious dysentery, pneumonia, enteritis, abscesses and mastitis caused by microorganisms sensitive to sulfametazim and trimethoprim.
Dogs, cats: treatment of animals suffering from pulmonary infections, septicemia, bacterial enteritis, coccidiosis in dogs caused by microorganisms sensitive to sulfametazim and trimethoprim.
Dosage
Intramuscularly at a dose of 1 ml per 10 kg of body weight of the animal daily for 3-5 days. The first day of treatment, the daily dose is divided into two parts and administered at intervals of 12 hours.
Contraindications
Hypersensitivity to sulfonamides. Do not administer to animals with renal or hepatic impairment. Do not use in conjunction with folates and p-aminobenzoic drugs.
Warning
Slaughter of animals for meat is allowed 28 days after the last use of the drug. The consumption of milk for human consumption is allowed 4 days after the last use of the drug. The meat and milk received before the specified period is disposed of or fed to unproductive animals, depending on the conclusion of a veterinary doctor.
Release form
Bottles made of transparent brown hydrolytic glass, closed with a rubber stopper for aluminum rinsing, 100 ml each. Secondary packaging – cardboard box.
Storage
A dry, dark place out of the reach of children at a temperature of 5 ° to 25 ° C.
The shelf life is 5 years.
For use in veterinary medicine!
Questions and answers of the customers
Related products
TYCLOSIN 500 mg/g Soluble Powder for dogs, chickens and pigs (100g/0.22lbs)
Tylosin (Tylan powder) 500 mg for chickens ● poultry ● pigs ● dogs
Tylosin is an antibiotic typically used to treat bacterial infections in farm animals, but veterinarians often use it to treat certain types of chronic diarrhea in cats and dogs. The powder form is not FDA approved for use in companion animals, but it is common practice for veterinarians to prescribe this medication. The drug is also given as an injectable type.
It is prescribed for the prevention and treatment of respiratory mycoplasmosis of birds, infectious sinusitis of turkeys, as well as the treatment of dysentery and gastroenterocolitis of bacterial etiology of pigs, bronchopneumonia of calves caused by pathogens sensitive to tylosin. To combat necrobacteriosis in cattle and small ruminants.
Enrofox 5% Solution for Cattle, Sheep, Cats, Pigs, Dogs (100ml)
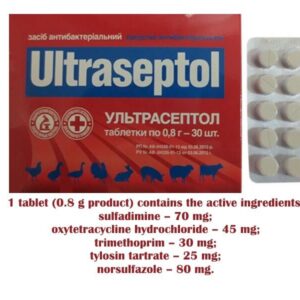
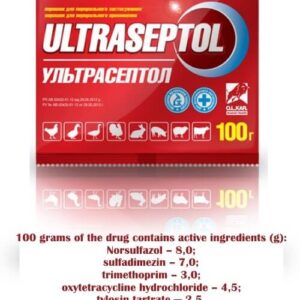
Ultrasept for Cattle, Pigs, Lambs, Rabbits, Poultry (100 tablets)
Ultrasept (sulfadimine, oxytetracycline, trimethoprim, tylosin, norsulfazole)
(Treatment of animals and poultry for diseases of the gastrointestinal tract, respiratory system and urogenital system caused by gram-positive, gram-negative and other microorganisms)
Calves under 3 months: treatment of animals suffering from gastroenteritis, salmonellosis, pasteurellosis, campylobacteriosis (vibriosis), digestive disorders with clinical signs of diarrhea caused by pathogens susceptible to the components of the preparation.
Pigs: Treatment of animals suffering from enzootic pneumonia, arthritis, dysentery, edema disease, erysipelas, salmonellosis, pasteurellosis, caused by microorganisms sensitive to components of the drug.
Lambs under 3 months: treatment of animals suffering from eymerioz, septicemia, contagious agalactia, caused by microorganisms sensitive to components of the drug.
Rabbits: the treatment of animals suffering from coccidiosis, pasteurellosis, salmonellosis, septicemia caused by pathogens susceptible to the components of the drug.
Poultry (chickens (broilers), turkeys, geese, ducks): treatment of birds suffering from coccidiosis, typhoid, cholera, salmonellosis, mycoplasmosis, rhinitis, caused by microorganisms sensitive to components of the drug.
Tiloxon powder For Broiler chickens, Poultry, Cattle (10g)
Doxycycline hydrochloride, tylosin tartrate, vitamins
For the treatment of mixed infections in young cattle, pigs and poultry, such as diseases of the gastrointestinal tract, respiratory tract, genitourinary system, skin, mastitis, metritis, agalactia.
The drug improves the growth and development of a young organism, reduces the likelihood of stress.
The drug is administered orally in a mixture with water or feed in the following doses:
poultry – at the rate of 1-1.5 g of preparation per 1 liter of drinking water;
young cattle – 1-1.5 g per 10 kg of body weight
pigs – 1-2 g per 10 kg of body weight.
For treatment, the drug is given twice a day for 3-5 days until the recovery or improvement of the general condition. With the preventive purpose – once a day for 3-5 days.
Tiloxon For Broiler chickens, Poultry, Cattle (1000g)
Doxycycline hydrochloride, tylosin tartrate, vitamins
For the treatment of mixed infections in young cattle, pigs and poultry, such as diseases of the gastrointestinal tract, respiratory tract, genitourinary system, skin, mastitis, metritis, agalactia.
The drug improves the growth and development of a young organism, reduces the likelihood of stress.
The drug is administered orally in a mixture with water or feed in the following doses:
poultry – at the rate of 1-1.5 g of preparation per 1 liter of drinking water;
young cattle – 1-1.5 g per 10 kg of body weight
pigs – 1-2 g per 10 kg of body weight.
For treatment, the drug is given twice a day for 3-5 days until the recovery or improvement of the general condition. With the preventive purpose – once a day for 3-5 days.
Ultrasept powder for Cattle, Pigs, Sheeps, Poultry 500 g
Ultrasept powder (sulfadimine, oxytetracycline, trimethoprim, tylosin, norsulfazole)
For the prevention and treatment of animals and poultry with primary and secondary bacterial infections of the respiratory, digestive and urinary tracts that is: the upper respiratory tract catarrh, pneumonia, enteritis, cystitis, mastitis, septicemia, postnatal complications, and a size of specific infections:
– Calves – Salmonella gastroenteritis kolibatselyarny, pasteurellosis, vibriosis;
– Pigs – enzootic pneumonia, arthritis, dysentery, edematous disease erysipelas, salmonellosis, pasteurellosis;
– Sheep – septicemia, eymerioz, contagious agalactia;
– Poultry – typhoid, cholera, salmonellosis, mycoplasmosis, rhinitis.
Metron powder for ornamental, aquarium fish and farm animals 500g (1.1 lbs)
Metronidazole 25% Powder (Metro Zole)
Exerts a bactericidal action on gram-positive and some gram-negative bacteria. It has a good effect against a size of pathogenic bacteria associated with ornamental fish diseases. In other words, diseases including Aeromonas and pseudomonas genera and mycobacterial group (gill diseases, Rhodococcus). It is useful in the control of “Hole-in-the-Head” and “Angel Problems” in Cichlid and Discus.
Treatment of cattle, sheep, pigs, fur-bearing animals, dogs, cats and birds suffering from trichomoniasis, dysentery, diarrhea, colitis, stomatitis, as well as diseases of the respiratory system, genitourinary system, gastrointestinal tract and skin caused by microorganisms sensitive to metronidazole. Dosage Orally with food in a dose of 10-20 mg of metronidazole per 1 kg m.t. for the active substance - twice a day for 5-7 days. If necessary, treatment is repeated after 7-8 days.