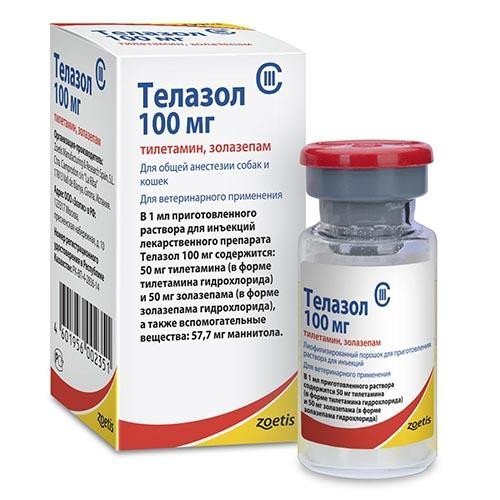
Telazol (tiletamine 50 mg, zolazepam 50 mg) Powder for injection
Telazol is an effective and safe combination drug for general anesthesia in diagnostic procedures and surgical interventions in dogs and cats.
Storage
Powder for solution for injection.
The reconstituted solution for injection contains 1 ml as active ingredients: 50 mg tiletamine (as tiletamine hydrochloride), 50 mg zolazepam (as zolazepam hydrochloride) and 57.7 mg mannitol as excipient. In appearance, the powder is a freeze-dried mass from white to amber. After dilution, the drug is a clear solution from colorless to slightly amber.
Telazole is available in the form of freeze-dried powder, packaged in glass vials with a capacity of 10 ml, sealed with rubber stoppers, reinforced with aluminum caps. The bottle is packed in a cardboard box. Instructions for its use are placed in each box with the drug.
Pharmacological properties
Telazole is a complex anesthetic.
tiletamine hydrochloride is an anesthetic with dissociative action, has a pronounced analgesic effect, but does not cause sufficient muscle relaxation. Teletamine hydrochloride does not suppress swallowing, laryngeal, cough reflexes, does not suppress the respiratory center.
Zolazepam hydrochloride is a benzodiazepine tranquilizer that inhibits subcortical areas of the brain, causing anxiolytic and sedative effects, as well as skeletal muscle relaxation. Zolazepam hydrochloride enhances the anesthetic effect of tiletamine hydrochloride. It also prevents the onset of seizures caused by tiletamine hydrochloride, improves muscle relaxation, and accelerates recovery from anesthesia.
The time of anesthesia after intramuscular administration of Telazol is individual, so after injection of the drug should be established careful monitoring of the animal.
In cats, the onset of action of the drug after intramuscular injection is observed after 1.5-6 minutes. The recovery period after anesthesia varies from 1 to 1.5 hours.
In dogs, the onset of action of Telazol after intramuscular injection is observed in 7-8 minutes. The recovery period is up to 4 hours.
The half-life of tiletamine in the blood plasma of dogs is 1.2 hours, zolazepam – 1 hour. In cats, the half-life of tiletamine is 2.5 hours, zolazepam – 4.5 hours.
The active substances of Telazole are metabolized in the liver and excreted in the urine. According to the degree of impact on the body Telazole is a moderately dangerous substance.
Indication
Telazole is used in dogs and cats for general anesthesia during diagnostic procedures and surgeries.
Doses and method of application
It is recommended to follow a fasting diet for at least 12 hours before using Telazole.
15 minutes before the use of Telazol for premedication, it is recommended to administer subcutaneously atropine sulfate in a dose: dogs – 0.1 mg / kg body weight, cats – 0.05 mg / kg body weight.
Immediately before use, 5 ml of water for injections with asepsis is injected into the vial of the drug. Water for injections is not part of the drug. The concentration of the resulting solution is 100 mg of active substances in 1 ml of the drug.
Telazole is used deep intramuscularly in the following indications in doses:
Dogs:
- Clinical examination – 7-10 mg / kg;
- Short-term general anesthesia for minor surgeries – 10-13 mg / kg;
- Prolonged general anesthesia for major surgery – no more than 25 mg / kg.
The maximum allowable dose is 29 mg / kg.
Cats:
- Clinical examination – 10-12 mg / kg;
- Short-term general anesthesia for minor surgeries – 10.5-12.5 mg / kg;
- Prolonged general anesthesia for major surgery – 14-16 mg / kg.
The maximum allowable dose is 72 mg / kg.
*Kilograms to a Pounds conversion table
| Kilograms (kg) | Pounds (lb) | Pounds+Ounces (lb+oz) |
| 0.1 kg | 0.220 lb | 0 lb 3.527 oz |
| 1 kg | 2.205 lb | 2 lb 3.274 oz |
| 5 kg | 11.023 lb | 11 lb 0.370 oz |
| 10 kg | 22.046 lb | 22 lb 0.740 oz |
Side effects
Apnea may occur with high doses or intolerance to Telazole 100 mg. During awakening, vomiting, hypersalivation, and bronchial hypersecretion may occur if atropine premedication has not been performed prior to anesthesia. During surgery – involuntary muscle twitching, hypertension, cyanosis, muscle rigidity.
In case of overdose, artificial lung ventilation is performed. Other symptoms are relieved by symptomatic therapy.
In case of individual intolerance of the components of the drug and the manifestation of allergic reactions, the drug should be discontinued.
Contraindication
Individual hypersensitivity to the components of the drug.
Telazole is contraindicated in animals with diseases of the pancreas, with severe disorders of the cardiovascular and respiratory systems, hypertension. In animals with kidney disease, the dose should be reduced. It is forbidden to use in pregnant animals, as well as in caesarean section, as the drug penetrates the placenta and can cause respiratory depression.
Special instructions
Concomitant use of Telazole and phenothiazine causes respiratory and cardiac depression, hypotension, hypothermia and pulmonary edema.
The drug is not intended for use in productive animals.
Personal prevention measures
When working with the drug should follow the general rules of personal hygiene and safety, provided when working with drugs. Wash hands with warm soapy water after use. In case of accidental contact of the drug with the skin or mucous membranes of the eyes, they should be rinsed with plenty of water. People with hypersensitivity to the components of Telazole should avoid direct contact with the drug. In case of allergic reactions or accidental ingestion of the drug, you should immediately contact a medical institution (have instructions for use or label). Empty vials from under the drug are disposed of with household waste.
Storage conditions
In a closed package of the manufacturer, in a dry, protected from light, out of reach of children and animals, separate from food and feed at a temperature of 20 C to 25 ° C. The shelf life of the drug in compliance with the conditions of storage in a closed package is 2 years from the date of manufacture. After dissolving the drug, the finished solution can be stored for 4 days at room temperature and for 14 days in the refrigerator.
Only clear solutions should be administered. Do not use the drug after the expiration date.
Unused drug with expired expiration date is disposed of in accordance with the requirements of the law.






































































Reviews
There are no reviews yet.