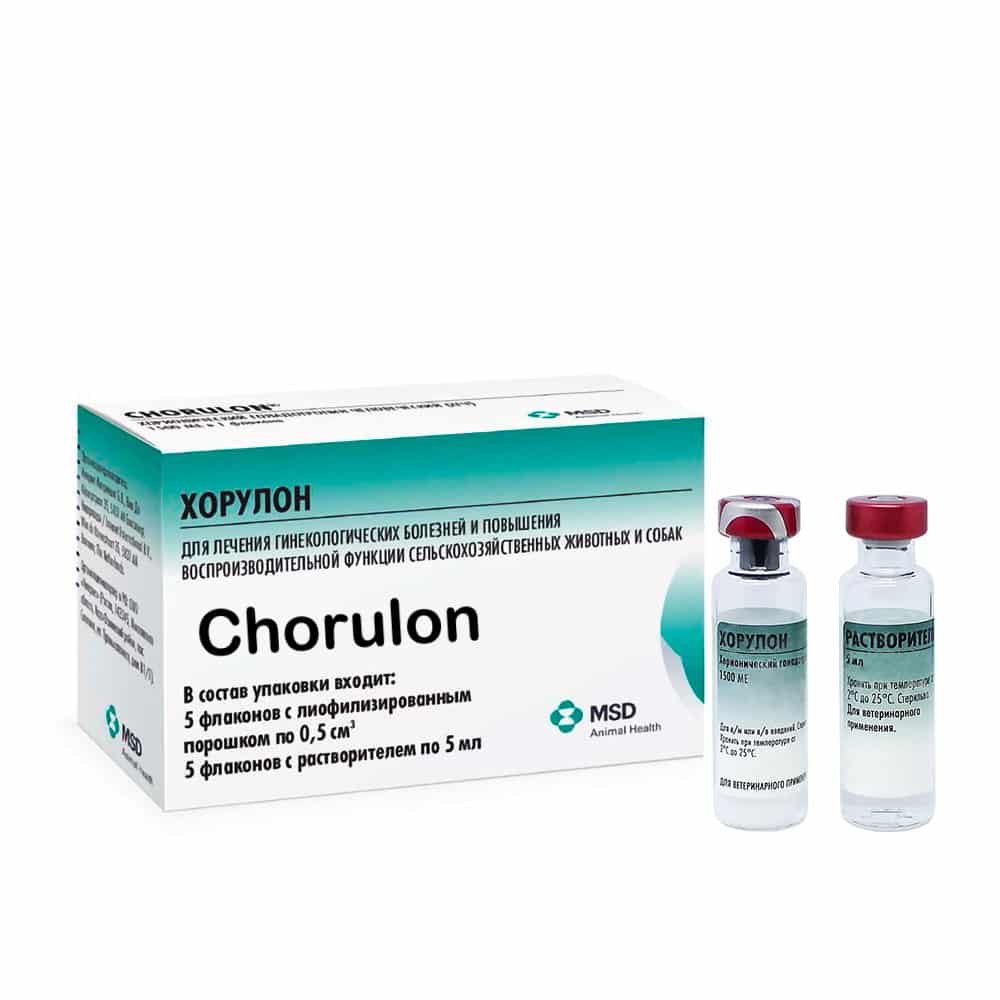
Chorulon (gonadotropin) solution, 5 ml
Trade name of the veterinary medicinal product: Chorulon®.
International non-proprietary name – chorionic gonadotropin.
Dosage form: lyophilized powder for solution for injection and solvent.
In appearance, the drug is a white powder, the solvent is a clear colorless liquid.
Storage
1 vial of lyophilized powder contains: as an active substance 1500 IU of hum chorionic gonadotropin (HCG), as excipients: mannitol, sodium hydrogen phosphate dihydrate, sodium dihydrogen phosphate dihydrate.
The composition of the solvent includes: sodium hydrogen phosphate dihydrate, sodium dihydrogen phosphate dihydrate, water for injections.
Pharmacological properties
Pharmacotherapeutic group: hormones and their antagonists.
Chorionic gonadotropin (HCG) acts on the ovary similarly to luteinizing hormone (LH) and binds to LH receptors. Horulon in females stimulates the function of interstitial ovarian cells, which ensure the maturation of the follicle, causes ovulation, promotes luteinization of granulosa cells and supports the activity of the corpus luteum, as well as increases the level of progesterone in the blood. In males, it stimulates testosterone production.
When injecting the drug, due to the high bioavailability of gonadotropic hormone (HCG), its maximum concentration in the blood plasma of animals is reached 8 hours after administration.
The elimination half-life is 10 hours in cows and 27 hours in pigs.
Horulon according to the degree of impact on the body is a low-risk substance (hazard class 4 according to GOST 12.1.007-76).
The order of application
Horulon is used to treat gynecological diseases, stimulate reproductive function and synchronize ovulation, treat follicular cysts, maintain corpus luteum function and blood progesterone levels, and treat nymphomania in female farm animals and dogs. In dogs, Horulon stimulates testosterone production and thus affects the development and maintenance of primary and secondary sexual characteristics.
Before use, the contents of the vial are dissolved at a rate of 1500 IU per 5 ml of solvent. The resulting solution is administered intramuscularly or intravenously.
The dose and method of administration depend on the species of animal and the indications for use and are given in the table
| View
animals |
Indications for use | Dose | Method of administration |
| Cows,
heifers |
To stimulate ovulation, increase reproducibility | 1500 IU, once, 2-3 hours before or simultaneously with fertilization | intramuscularly or intravenously |
| Prevention of embryonic mortality | 1500 IU, once in 6 days after fertilization | intramuscularly | |
| Treatment of ovarian follicular cysts, anestrus, prolonged hunting, nymphomania | 3000 IU, once | intravenously | |
| Mares | To stimulate ovulation (follicles> 3.5 cm) | 1500 – 3000 IU, once, 24 hours before insemination | intramuscularly or intravenously |
| Anestrus (follicles> 2 cm) | 1500 – 3000 IU, once, 24 hours before or at the time of fertilization; if necessary, re-injected after 48 hours | intramuscularly or intravenously | |
| Sows,
repair pigs |
Stimulation and synchronization of ovulation | 500 IU once, 24 hours before or during the incident | intramuscularly |
| Bitches | Anestrus | 500 IU, once on the first day of heat after previous treatment with Folligon | intramuscularly or intravenously |
| Delayed ovulation, prolonged heat | 100 – 800 IU, once a day, repeat the treatment until the vaginal discharge disappears (but not more than 5-8 days) | intramuscularly | |
| Males | Cryptorchidism | 100 – 500 IU, 2 times a week for 6 weeks | intramuscularly |
| Lack of libido | 100 – 500 IU, once, 6-12 hours before mating | intramuscularly |
*Kilograms to a Pounds conversion table
| Kilograms (kg) | Pounds (lb) | Pounds+Ounces (lb+oz) |
| 0.1 kg | 0.220 lb | 0 lb 3.527 oz |
| 1 kg | 2.205 lb | 2 lb 3.274 oz |
| 5 kg | 11.023 lb | 11 lb 0.370 oz |
| 10 kg | 22.046 lb | 22 lb 0.740 oz |
Contraindication
It is forbidden to use Horulon in case of hypersensitivity of the animal to the components of the drug.
It is forbidden to use the drug Horulon during pregnancy more than 2 weeks. The use of Horulon is allowed during the first 2 weeks of pregnancy, as well as lactating animals.
Side effects
In rare cases, anaphylactic reactions are possible with the use of the drug Horulon. In these cases, immediate administration of adrenomimetics (1: 1000) intravenously or intramuscularly is recommended.
Overdose
Symptoms of overdose with the use of the drug in accordance with these instructions have not been established.
Features of application
There is no information on the peculiarities of the use of Horulon with drugs of other pharmacological groups.
Violations of the timing of the drug should be avoided, as this may reduce the therapeutic efficacy of treatment. If you miss the next injection, you must use the drug as soon as possible in the same dose.
Features of action
Features of action at the first reception of drug and its cancellation are not revealed.
Withdrawal period
Meat, slaughter products after the use of the drug Horulon can be used for food purposes without restrictions.
Safety and personal hygiene measures
When working with Horulon should follow the general rules of personal hygiene and safety, which are provided when working with drugs.
People with hypersensitivity to the components of the drug should avoid direct contact with Horulon. Do not drink, smoke or eat while working with the drug; after work, wash your hands with warm soapy water.
It is forbidden to use empty vials of the drug for household purposes, they must be disposed of with household waste.
In case of accidental contact of the drug with the skin or mucous membranes, they should be rinsed with plenty of clean water. In case of allergic reactions or accidental ingestion of the drug should immediately contact a medical institution (have instructions for use of the drug or label).
Release form
Horulon is released in packages of 1500 IU HCG (0.5 cm) in glass vials with a capacity of 5 ml. The solvent is packed in 5 ml glass vials of appropriate capacity. Vials with lyophilized powder and solvent are hermetically sealed with rubber stoppers reinforced with aluminum caps. 5 vials of lyophilized powder and 5 vials of solvent are packed in cardboard boxes with instructions for use.
Storage
Horulon is stored and transported at a temperature of 2 to 25 ° C. Horulon is stored out of reach of children.
Expiration date
Shelf life of the drug Horulon in compliance with the conditions of storage and transportation – 3 years from the date of manufacture. After dissolution, store the drug at a temperature of 2 to 8 ° C, use within 24 hours.
Do not use the drug after the expiration date.
Unused drug is disposed of in accordance with legal requirements.
Horulon is available without a prescription from a veterinarian.